Water quality standards are the backbone of safe drinking water systems. They define which contaminants matter, how much of each substance can be present, how utilities should monitor water, and when treatment or public notification is required. For households, utilities, regulators, and public health professionals, understanding these standards helps turn a complex scientific topic into something practical: is the water safe to drink, what do test results mean, and what should happen if limits are exceeded? In this article, we explain how water quality standards work, how the World Health Organization and the U.S. Environmental Protection Agency approach them, why numbers differ across jurisdictions, and how these standards connect to water testing, treatment, and everyday health protection.
What are water quality standards?
In the drinking water context, water quality standards are formal health-based or regulatory benchmarks that set acceptable concentrations for microbiological, chemical, and physical parameters in water intended for human consumption. These benchmarks may be called guidelines, standards, maximum contaminant levels, action levels, treatment technique requirements, or health advisory values depending on the organization and legal system involved.
At a basic level, a water quality standard answers one or more of these questions:
- Which contaminants should be monitored?
- What concentration is considered acceptable or tolerable?
- How often should water be tested?
- What analytical methods should be used?
- What treatment is required if contamination is found?
- When must the public be informed?
Not all standards are legally identical. Some are enforceable regulations. Others are guideline values intended to support national decision-making. This distinction is central to comparing WHO water guidelines and EPA drinking water standards.
Why water quality standards matter for drinking water safety
Without water quality standards, water safety regulation would be inconsistent, reactive, and often too late to prevent harm. A contaminant may be invisible, tasteless, and odorless while still posing substantial health risk. Standards create a common scientific framework for prevention. They guide source protection, treatment design, distribution system management, surveillance, and consumer communication.
They matter because drinking water can contain hazards from many sources:
- Pathogens from sewage, animal waste, or treatment failures
- Metals such as lead and arsenic from geology, industrial contamination, or plumbing materials
- Nutrients such as nitrate from agriculture, septic systems, or manure
- Disinfection byproducts formed during water treatment
- Industrial chemicals, solvents, and emerging contaminants
- Natural substances such as fluoride, manganese, or radionuclides
Standards also support proportional response. A bacterial contamination problem may require boil water notices and immediate corrective action, while a slowly accumulating contaminant such as arsenic may trigger treatment upgrades and long-term compliance planning. The number alone is not the entire story; the hazard type, exposure duration, and vulnerable population all influence the public health response.
For readers who want broader context on contaminant categories and treatment pathways, PureWaterAtlas provides a useful overview of water science, contaminants, treatment, and water quality.
The scientific foundation behind standards
Scientific water quality standards are not arbitrary numbers. They are derived from toxicology, epidemiology, microbiology, exposure science, analytical chemistry, and engineering feasibility. Regulators and international agencies assess how contaminants behave, what health effects they can cause, who is most vulnerable, and how often people may be exposed.
Hazard identification
The first step is determining whether a contaminant can cause harm. For microbes, this often involves documenting infectious dose, routes of transmission, outbreak evidence, and survival in water. For chemicals, hazard identification relies on animal studies, human epidemiological data, mechanistic studies, and case reports.
Dose-response assessment
This step evaluates how risk changes with exposure level. Some substances have threshold effects, meaning low enough exposures are not expected to cause harm. Others, especially some carcinogens, may be treated using models that assume some level of risk even at very low exposure. This difference helps explain why some drinking water limits are set with a margin of safety well below concentrations known to cause obvious illness.
Exposure assessment
Risk depends on how much water people drink, how long they are exposed, whether they are also exposed from food or air, and whether special populations are affected differently. Infants, pregnant people, immunocompromised individuals, and people with certain kidney or metabolic conditions may be more susceptible to some contaminants.
Risk characterization
Agencies combine hazard, dose-response, and exposure information to estimate risk and derive health-based targets. These targets may then be adjusted to reflect practical realities such as analytical detection limits, treatment capability, monitoring logistics, and cost-benefit considerations, depending on the regulatory framework.
Microbiological risk assessment
Pathogen-related standards often differ from chemical standards because microbial contamination can cause acute illness quickly and outbreaks can spread rapidly. Instead of relying only on numerical concentration limits for every pathogen, drinking water systems often use indicator organisms, treatment performance requirements, and multiple barrier approaches. For example, a utility may be required to achieve specific levels of pathogen removal or inactivation through filtration and disinfection, rather than simply meet an end-point number for each organism.
WHO water guidelines: what they are and how they work
The WHO water guidelines are global public health recommendations designed to help countries establish or revise national drinking water regulations and risk management systems. The World Health Organization does not directly regulate municipal water utilities in most countries. Instead, it provides evidence-based guideline values and a broader framework for ensuring safe drinking water from catchment to consumer.
The WHO emphasizes preventive risk management, including water safety plans, source protection, treatment control, operational monitoring, and verification testing. Its approach recognizes that end-product testing alone is not enough. A utility can produce compliant test results on many days and still experience a contamination event if system controls fail.
The WHO’s drinking water information is summarized in its public health materials on drinking water safety and health, while the full guideline framework is detailed in the Guidelines for Drinking-water Quality.
Guideline values versus regulations
A key point is that WHO guideline values are generally advisory, not automatically enforceable law. Countries may adopt them directly, modify them, or create stricter or more flexible national standards based on local conditions. That does not make them less scientific. It means they are designed for broad applicability across nations with very different water sources, treatment infrastructure, climates, and public health burdens.
Water safety plans
One of the WHO’s most influential contributions is the water safety plan concept. This is a comprehensive risk assessment and risk management framework covering the entire supply chain:
- Protection of source waters
- Control of treatment processes
- Maintenance of storage and distribution systems
- Operational monitoring of critical control points
- Verification through laboratory testing
- Documentation, training, and incident response
This systems approach is especially important for microbial safety. If a treatment barrier fails, waiting for finished-water test results may mean consumers have already been exposed.
Health-based targets
WHO uses several types of health-based targets depending on the hazard and setting. These may include a specific concentration guideline, a performance target for treatment, or a risk-based metric. This flexibility reflects the reality that safe water delivery is both a scientific and operational challenge.
EPA drinking water standards: what they are and how they work
In the United States, EPA drinking water standards are established primarily under the Safe Drinking Water Act. Unlike WHO guidelines, many EPA standards are enforceable for public water systems. The EPA sets legally binding maximum contaminant levels, treatment technique requirements, monitoring rules, and reporting obligations. States may implement and enforce these requirements if they meet federal criteria, and some states adopt stricter rules than the federal baseline.
The EPA’s central drinking water regulatory portal is available through its ground water and drinking water program.
MCLGs and MCLs
The EPA framework often distinguishes between:
- Maximum Contaminant Level Goals (MCLGs): non-enforceable health-based goals set at a level at which no known or anticipated adverse health effect would occur, with an adequate margin of safety
- Maximum Contaminant Levels (MCLs): enforceable limits set as close to the MCLG as feasible, considering treatment technology and cost
This distinction is important because the health ideal and the regulatory limit are not always identical. For some contaminants, especially carcinogens or lead, the ideal health-based goal may be zero, even though the enforceable standard uses a practical regulatory framework.
Treatment techniques and action levels
Not all hazards are regulated through a single concentration limit in finished water. For some contaminants, the EPA uses treatment technique rules or action levels. The Lead and Copper Rule is the classic example: compliance is based on household sampling and a percentile-based action level rather than a simple universal MCL in all taps. This reflects the fact that lead contamination often arises in plumbing after water leaves the treatment plant.
National Primary and Secondary Drinking Water Regulations
EPA drinking water standards include two broad categories:
- Primary regulations: enforceable standards for contaminants that may affect health
- Secondary regulations: non-enforceable guidelines for contaminants affecting taste, odor, color, staining, or cosmetic characteristics
Secondary parameters such as iron, manganese, chloride, sulfate, or total dissolved solids may not always present a direct health risk at typical levels, but they still matter because they influence acceptability, plumbing performance, and consumer confidence.
WHO versus EPA: why the numbers are not always the same
People often assume that if two respected authorities list different values for the same contaminant, one must be wrong. In reality, differences can arise for legitimate scientific and policy reasons.
Different legal roles
WHO develops global guidance. EPA develops national, enforceable U.S. rules. Guidelines can be purely health-based, while regulations may incorporate feasibility, monitoring design, and system-wide implementation constraints.
Different assumptions and update cycles
Agencies may review evidence at different times, use different toxicological endpoints, assign different uncertainty factors, or weigh epidemiological studies differently. As science evolves, standards can change. Regulatory updates often lag behind emerging evidence because rulemaking requires formal review and implementation planning.
Different local conditions
A nation with widespread groundwater fluoride deficiency, arsenic-rich geology, limited treatment infrastructure, or a high burden of diarrheal disease may make different regulatory choices from a highly industrialized country with advanced centralized treatment systems. Water policy is always scientific, but it is also contextual.
Different risk management strategies
For microbial hazards, one framework may emphasize treatment performance and sanitary inspection, while another may rely more heavily on specific compliance indicators. For chemical hazards, one system may prioritize achievable numerical standards while another emphasizes health-based targets and locally adapted management.
Main categories of contaminants covered by water quality standards
Microbiological contaminants
These are the most immediate threats to drinking water safety. Pathogens include bacteria, viruses, and protozoa such as:
- Escherichia coli and fecal indicator bacteria
- Salmonella
- Norovirus and other enteric viruses
- Giardia
- Cryptosporidium
Microbiological standards often focus on indicators because testing every sample for every pathogen is impractical. Total coliforms, E. coli, turbidity, disinfectant residuals, and treatment performance metrics are commonly used to infer system integrity and pathogen control.
Inorganic chemicals
This group includes metals, metalloids, and ions such as arsenic, lead, copper, nitrate, nitrite, fluoride, selenium, uranium, and chromium. Some occur naturally in groundwater. Others result from corrosion, mining, manufacturing, agriculture, or waste disposal.
Organic chemicals
These include pesticides, industrial solvents, petroleum components, and disinfection byproducts. Their occurrence varies with land use, industrial history, source water type, and treatment chemistry.
Radiological contaminants
Radium, uranium, gross alpha activity, and other radionuclides are regulated in many systems because long-term ingestion may increase cancer or kidney-related risks depending on the contaminant.
Aesthetic and operational parameters
pH, hardness, turbidity, conductivity, total dissolved solids, iron, manganese, taste, odor, and color are not merely cosmetic details. They can signal treatment performance, corrosion conditions, source changes, or the potential for broader water quality issues.
Examples of important drinking water limits
The phrase drinking water limits usually refers to concentrations above which a contaminant is considered unsafe, noncompliant, or in need of response. However, the exact meaning depends on the parameter.
| Parameter | Why it matters | Type of standard commonly used |
|---|---|---|
| E. coli | Indicates fecal contamination and possible pathogen presence | Typically zero tolerance in distribution samples intended for compliance |
| Nitrate | Can cause serious health risk in infants at elevated levels | Numerical maximum contaminant level or guideline value |
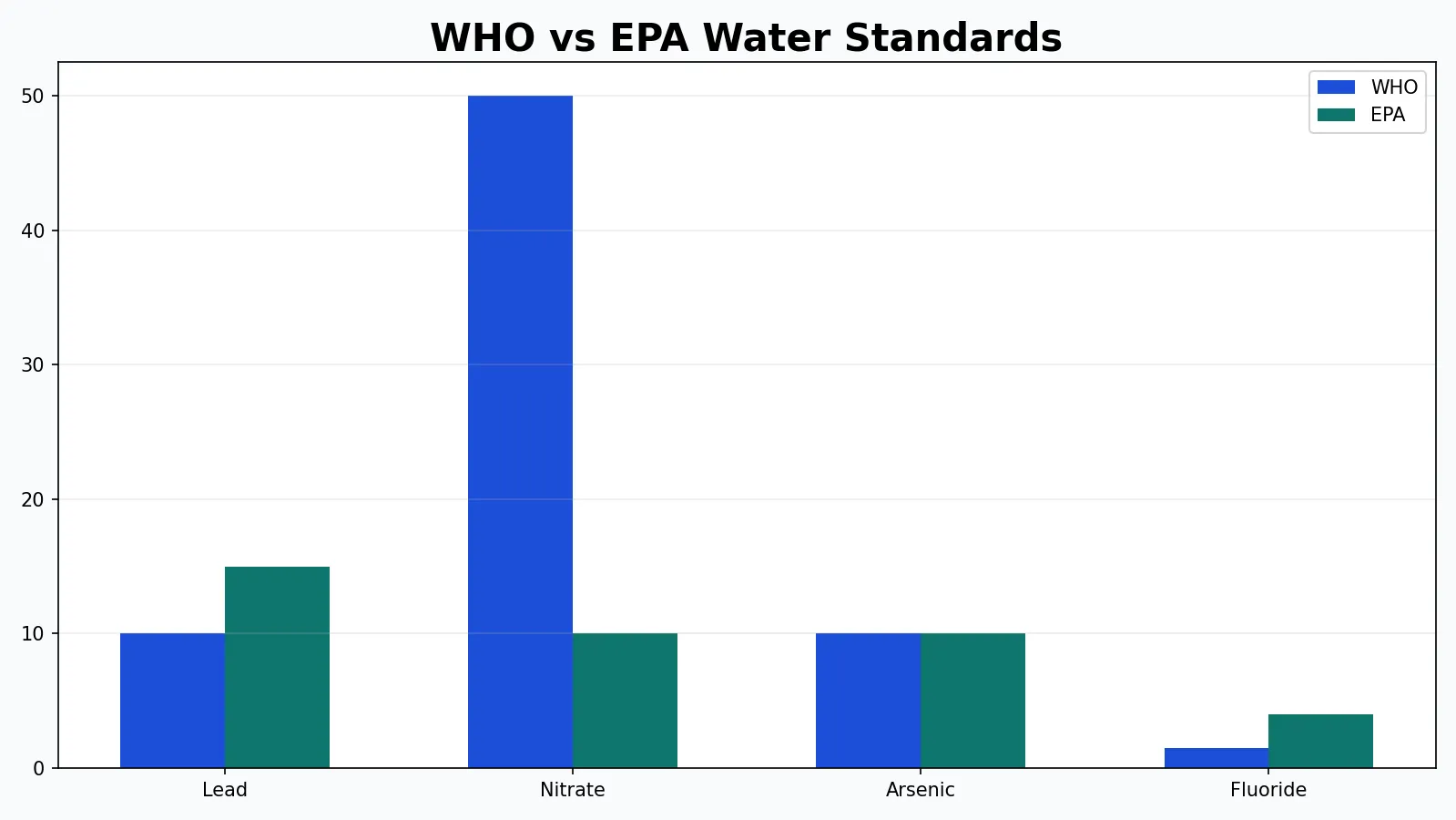
| Lead | Neurotoxic, especially harmful to children; often enters water from plumbing | Action level and corrosion control requirements in some regulatory systems |
| Arsenic | Associated with long-term toxic and carcinogenic effects | Numerical maximum contaminant level or guideline value |
| Turbidity | Can interfere with disinfection and signal treatment failure | Operational and treatment performance requirement |
| Total trihalomethanes | Disinfection byproducts linked to chronic risk concerns | Running annual average or similar compliance metric |
Even when consumers learn a number, interpretation should account for sampling location, time basis, laboratory method, and whether the benchmark is a health goal, enforceable limit, operational trigger, or advisory value.
Lead, nitrate, arsenic, and microbes: four contaminants that show how standards work in practice
Lead
Lead is a textbook example of why water quality standards must account for both toxicology and plumbing. Lead usually does not originate in source water at high concentrations; it more often enters water through corrosion of service lines, solder, and brass fixtures. Because household-specific plumbing conditions matter, compliance sampling is challenging.
Health agencies generally agree that there is no beneficial level of lead exposure. Children and fetuses are especially vulnerable. Yet regulatory systems may use an action level rather than an absolute concentration standard at every tap because the risk arises within the distribution and premise plumbing environment. Homeowners concerned about this issue can explore practical sampling considerations in PureWaterAtlas’s guide to testing lead in water.
Nitrate
Nitrate contamination is strongly associated with fertilizers, manure, septic systems, and agricultural runoff. It is especially important in private wells and shallow groundwater. Elevated nitrate can impair oxygen delivery in infants, causing methemoglobinemia, sometimes called blue baby syndrome. This is why nitrate has long been one of the most important drinking water limits in groundwater-dependent communities.
If nitrate is a concern in your area, especially for well users, see PureWaterAtlas’s article on testing nitrates in water.
Arsenic
Arsenic often occurs naturally in bedrock and aquifers, though industrial sources can also contribute. Chronic exposure is associated with increased risks of cancer and other health effects. Arsenic illustrates how a contaminant can be completely invisible while still requiring highly specific laboratory testing and specialized treatment such as adsorption, coagulation-filtration, or reverse osmosis.
Microbial contamination
Microbial hazards differ from lead, nitrate, and arsenic because the risk may be acute, immediate, and outbreak-driven. A single failure in source protection, disinfection, or distribution integrity can expose many people quickly. Standards therefore emphasize prevention, operational control, and rapid response. Public health guidance from the CDC’s drinking water resources helps explain why boil water notices and emergency communication are essential parts of safe water management.
How standards are measured: sampling and laboratory testing
A standard is only meaningful if water can be measured accurately. Drinking water testing depends on representative sampling, contamination-free collection, validated analytical methods, and correct interpretation.
Sampling location matters
Results can differ dramatically depending on where the sample is taken:
- At the source water intake or well
- After treatment
- Within the distribution system
- At the consumer’s tap
For lead, first-draw household samples may be highly informative. For microbial compliance, distribution system samples are critical. For treatment plant optimization, multiple in-process samples may be needed.
Time matters
Some standards are based on single-sample maximums. Others use averages over months or years. Seasonal variation can affect nitrate, pesticides, algal toxins, and microbial risk. Rainfall events may increase turbidity and pathogen loading. Warm temperatures can shift disinfectant demand and byproduct formation.
Method detection limits matter
If the lab cannot detect a contaminant at concentrations relevant to the standard, the result may be of limited value. Laboratories use specific validated methods for metals, ions, volatile organics, microbes, and radiological analytes. The method must be fit for purpose.
Field tests versus certified laboratory tests
Home test kits can be useful for screening parameters like pH, hardness, chlorine, and sometimes nitrate. However, regulatory comparison usually requires certified laboratory analysis, especially for lead, arsenic, pesticides, volatile organic compounds, and microbiological compliance.
For a broader overview of how household and lab-based analysis fit together, readers can browse the PureWaterAtlas water-testing category.
How to interpret water test results against standards
Many people receive a lab report and immediately look for a red flag number. That is understandable, but interpretation should be more careful. A result only becomes meaningful when compared with the correct benchmark and context.
Questions to ask when reviewing results
- Is the benchmark a WHO guideline, an EPA enforceable standard, a state value, or a health advisory?
- Was the sample taken from a tap, a well, or the treatment plant?
- Does the standard apply to a single sample or an average?
- What are the units: mg/L, µg/L, CFU, MPN, or something else?
- Was the contaminant detected above the reporting limit but below the regulatory limit?
- Is the result likely to vary over time?
For readers who want help decoding real reports, PureWaterAtlas offers a practical guide to understanding water test results.
Below the standard does not always mean zero risk
Regulatory compliance is important, but it does not mean a substance is absent. It means the concentration falls below the applicable benchmark and is considered acceptable within that framework. Sensitive individuals may still need targeted advice in some cases, and private well owners may need to make decisions based on local risk even without formal regulatory enforcement.
Above the standard does not always mean immediate poisoning
Exceedance should always be taken seriously, but the urgency depends on the contaminant. E. coli in drinking water can justify immediate avoidance and corrective action. A moderate exceedance of an aesthetic parameter may be less urgent. Chronic contaminants such as arsenic may pose long-term risk rather than immediate symptoms, but still require prompt planning for treatment or alternative water.
Public water systems versus private wells
One of the most misunderstood parts of water safety regulation is that regulatory oversight often applies primarily to public water systems, not private wells. In many countries, households using private wells are responsible for their own testing and treatment decisions.
This creates a major gap between standards on paper and exposure in real life. Public systems may provide annual consumer confidence reports, regular monitoring, and mandated treatment. Private well users may have none of those protections unless they act proactively.
Anyone unsure about their local situation should start with a practical overview of how to know if your tap water is safe to drink. That is especially important after floods, plumbing work, changes in taste or odor, or when vulnerable people are in the home.
Water policy and regulatory implementation
Water policy determines how standards move from scientific documents into daily practice. A strong standard is only effective if it is backed by monitoring capacity, trained operators, laboratory infrastructure, treatment funding, enforcement, and transparent public communication.
Key components of effective water safety regulation
- Clear legal authority and defined responsibilities
- Routine monitoring and reporting requirements
- Accredited or certified laboratory support
- Operator training and competency standards
- Source water protection programs
- Corrosion control and infrastructure maintenance
- Emergency response and boil water protocols
- Public notification rules and health communication
Good water policy also accounts for equity. Small systems, rural communities, and low-income regions may face the highest contamination risk while having the fewest resources for compliance. Standards should protect health without creating unrealistic burdens that lead to underreporting or nonimplementation.
How source water affects standards and compliance
Not all water sources present the same challenges. Groundwater is often lower in pathogens than surface water, but it may contain more arsenic, fluoride, manganese, uranium, or nitrate depending on geology and land use. Surface water is more vulnerable to runoff, microbial contamination, turbidity spikes, and algal blooms, but large utilities often have robust treatment barriers.
Hydrology, geology, and watershed activity strongly shape the contaminants that standards must address. The USGS water resources program provides valuable background on how water moves through natural systems and why source conditions matter for water quality.
Emerging contaminants and the future of standards
Water quality standards are not static. Scientific priorities change as new contaminants are detected, analytical methods improve, and toxicological evidence evolves. PFAS, microplastics, cyanotoxins, pharmaceutical residues, and antibiotic resistance markers have all increased attention to the limits of traditional regulatory frameworks.
Not every detectable substance will become a regulated contaminant. To justify a formal standard, agencies usually need evidence that the substance occurs in drinking water at concerning levels, poses meaningful health risk, and can be monitored reliably. Even then, translating science into regulation can take years.
This is why risk communication must be careful. “Detected” does not always mean “dangerous,” but absence of a standard does not automatically mean “safe.” The scientific process is iterative.
Common misconceptions about water quality standards
If my water meets standards, it is perfect
Not necessarily. It means the water meets the applicable benchmarks that were measured. There may still be taste, hardness, scaling, or localized plumbing issues. Also, some contaminants are not tested continuously at every tap.
If my utility report shows no violation, my home tap cannot have lead
Incorrect. Lead often comes from premise plumbing. A utility may be compliant while a specific home still has a lead service line or corrosive plumbing conditions.
WHO and EPA values should always be identical
No. Different legal roles, assumptions, update cycles, and local contexts can produce different but scientifically defensible values.
A home filter automatically makes water compliant with all standards
No. Filter performance depends on technology, certification, installation, flow conditions, maintenance, and the contaminant in question. Some filters improve taste only. Others target specific hazards such as lead, nitrate, or pathogens.
FAQ
What is the difference between WHO water guidelines and EPA drinking water standards?
WHO guidelines are international, science-based recommendations that help countries develop their own drinking water rules. EPA drinking water standards are U.S. federal regulatory requirements for public water systems and are enforceable under national law.
Are water quality standards the same everywhere?
No. Countries and states may use different values or implementation methods based on science reviews, legal frameworks, treatment feasibility, and local exposure conditions.
Do water quality standards apply to private wells?
Usually not in the same way they apply to public water systems. Private well owners are often responsible for testing and treatment themselves, even though the same health-based concerns still matter.
Which contaminants are most important to test for?
That depends on the water source and local risk. Common priorities include coliform bacteria or E. coli, nitrate, lead, arsenic, hardness, pH, manganese, and region-specific contaminants such as pesticides or uranium.
If a test result is above a standard, what should I do?
Confirm the result, review the sampling context, identify whether the issue is acute or chronic, and seek guidance from your water utility, local health department, or a qualified water treatment professional. For microbial contamination or boil water instructions, act immediately according to official advice.
Conclusion
Water quality standards translate complex science into practical protection for public health. Whether expressed as WHO water guidelines, EPA drinking water standards, action levels, treatment techniques, or national drinking water limits, they help define what safe water should look like and how systems must respond when quality is threatened. Understanding these standards is not just for regulators. It helps households interpret test results, ask better questions, recognize the difference between aesthetic and health concerns, and make informed decisions about testing, treatment, and ongoing monitoring. Safe drinking water depends on both sound science and strong implementation, and water quality standards are where those two goals meet.
Read the full guide: Water Testing Guide
Explore more in this category: Water Testing Articles